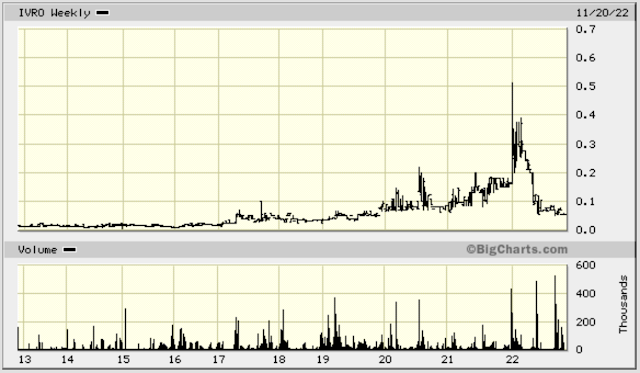
Sometimes a stock just looks so good you have to buy. InVitro International (IVRO) is tiny, profitable, and debt free. The stock sits at a low after falling from a big rise.
When assessing potential you have to start with the business. I don't go that deep with relative competitive analysis or anything but with a quick glance you can at least know whether you have a melting ice cube. IVRO is looking to the future. They are pushing the world towards non-animal based testing. Making products that allow development of lotions and cosmetics without involving animals. I believe in the mission and find it hard to believe the world would not move in this direction. A few quotes from the most recent annual:
The Company is a pioneer in the field of non-animal testing and was first to develop and commercialize its flagship product Corrositex® in 1991. The global regulatory bodies that govern non-animal testing did not exist at the time. These regulatory bodies started to evolve in early 2000’s and then consolidated into a more robust global regulatory system in the last few years. Organization for Economic Co-operation and Development (OECD) is the foremost such regulatory body today, with 37 member countries, including the US, and covers more than 80% of the world of commerce.Now to some numbers. My favorite thing about IVRO is how small and tight it is. In the market's opinion, a lot of houses are worth more.
In 2014, Corrositex® was adopted by OECD, with the publication of Test Guideline (TG) 435. Following this regulatory approval, Corrositex®, became Global Harmonization System (GHS) accepted as a full replacement for animal test results virtually everywhere in the world of commerce. The OECD/European Centre for the Validation of Alternative Methods (ECVAM), Transport Canada, U.S. DOT, EPA, OSHA, Consumer Product Safety Commission, FDA, and the International Air Transport Authority (IATA) all have accepted Corrositex® as an alternative as well.
In November 2019, the OECD published Test Guideline (TG) 496, the final step in the adoption of the Company’s now 30 year old core technology, Ocular Irritection® (OI). OI thus completes an eleven year effort to become the first 100% animal free ocular irritancy test method to be OECD accepted, validated and adopted for 37 countries, including the U.S.
Each of the above mentioned Regulatory Advancements are the result of many years in a strategic alliance with INT.E.G.RA in Italy. The Company partnership sells and distributes both laboratory test results and kits in Italy and several European markets. In addition, the partnership coordinates and facilitates regulatory approvals and acceptances from authorities and agencies within the OECD. The Company has over a dozen testing laboratory partnerships globally to help commercialize its technology.
- stock price $0.055
- market cap $1.25m
- 22.66m shares out
- book value $1.42m
- cash $1.17m
- debt zero
- revenue ~$1m
- earnings positive averaging $68k over past 4 years
- $0.003 EPS
- 35% inside ownership
- Aug 2017 IVRO starts putting out press releases filed with OTC markets new service with such exciting titles as "InVitro News Release 12.7.20"
- Feb 2018 management transition. Leader of 23 years is stepping down as president. New president is a former employee Kleiner.
- "Mr. Ulmer will maintain his position as CEO, along with his seat on the board, and begin putting greater emphasis on properly positioning InVitro International to take full advantage of developing opportunities in the global testing alternatives marketplace."
- June 2019 new president taking over Jhalani
- "Jhalani has joined the company as President to accelerate the company’s strategic objective of becoming the leading “in vitro” non-animal testing company globally"
- Dec 2019 the OECD announces IVRO product is adopted for global use
- "OI is the first 100% animal free ocular irritancy test method to be adopted by OECD and its 36 member countries representing about 80% of global trade, including the United States. This adoption culminates an 11 year effort with OECD which is viewed as the world’s foremost Regulatory body for in vitro test methods"
- "In 1985 IVRO pioneered 100% cruelty free test methods when there was not even one specific regulatory body to set standards or approve test methods anywhere in the world. Now 35 years later, OECD adoption represents the highest scientific review level for any non-animal test method"
- June 2021 IVRO hires an investor relations firm
- "With our continued financial improvements, recovering economy and improved regulatory condition, we believe InVitro presents an extremely undervalued investment story that will resonate with the micro-cap investment community"
- Aug 2021 IVRO starts their new add campaign "Mission to Help Make Non Animal Testing Famous" detailing out their strategy
- "In June, 2021, Maine became the sixth U.S. state to ban the sales of new cosmetic products tested on animals. It now joins Hawaii, California, Illinois, Nevada and Virginia in implementing such a ban; with impending legislation in New Jersey, Maryland, New York, Rhode Island and Oregon. Is a U.S. wide ban in our near future? China is the second largest cosmetics market in the world and its long standing requirement for 'animal testing' of cosmetic products has been a major hurdle for non-animal testing globally. In 2021, China provided a pathway for companies to import as well as to sell non animal tested cosmetics. This global legal momentum and changing consumer attitudes bodes well for our future.'"
- Sept 2021 IVRO announces a search for "strategic partners"
- "announced today that it has taken a first step in its search for strategic partners. IVRO entered into Letter of Intent (LOI) discussions where, although nothing certain can be forecasted, the Company believes results could be instrumental in furthering IVRO's primary mission to "Help Make Non-Animal Testing Famous""
- Dec 2021 IVRO gave an update on the legislative climate with annual results
- "This fiscal year has brought significant improvement in global laws for non-animal testing. China removed its requirements for animal testing for most imported cosmetic and personal care products. Mexico recently became the first country in North America and the 41st country in the world to ban the sales of new cosmetics tested on animals. In the United States, New Jersey became the eighth state in the country, and the fifth this year, to pass a law banning the sale of new cosmetics tested on animals, following California, Nevada, Illinois, Virginia, Maryland, Maine and Hawaii"
- Mar 2022 IVRO announces buying back 100k shares
- "I believe our decision reflects IVRO management's continuing strong belief in the future of NON-Animal testing; worthy of special note, just recently "The Humane Cosmetics Act of 2021" arrived on the floor of the U.S. Congress after many years in Committee"
- Nov 2022 IVRO announces a new opportunity for growth by buying a testing lab. And seems they want to buy more.
- "announced it has signed its first Letter of Intent (LOI) to acquire a privately owned, U.S. based testing laboratory. Over many years this laboratory has served IVRO targeted markets profitably and its principals agree that the future is bright for NON-Animal testing."
- "this initial acquisition effort may be viewed as a great opportunity by other laboratories globally. Acquisitions by public companies can provide an exit strategy for the owners/founders of privately owned laboratories, allowing them to continue to operate their businesses independently, as a wholly owned subsidiary of a public entity. This also provides potential growth opportunities and synergies for IVRO. We believe we can benefit from broader market exposure, faster development of in vitro test technologies, and increased revenue and resources.""
--Dan
disclosure: long IVRO
Subscribe to all NoNameStocks posts here: https://forms.feedblitz.com/erd
It looks like they're only profitable because of PPP loan forgiveness. But they do have no debt, at least.
ReplyDeleteIVRO has been profitable every year since 2014 which is the oldest financials posted on otcmarkets. Most recently yes it's been thanks to PPP
DeleteYou're right, my mistake. And it looks like the driver of their operating profit/loss is the R&D spend, which is where the PPP loans went, whereas their SG&A spend is more stable.
DeleteThis is a very interesting find of yours, Dan.
Hi Dan,
ReplyDeleteDo you think that at this moment at 8 cents it is to much to pay? Also do you use any filter to find ideas?
Well I've bought at 8 cents. I've bought lower and higher. I have never sold even when it ran to 0.50 and sat around 0.30. I'm not looking for pennies. But that's just me, you need to figure out for yourself
DeleteAs to a filter no I don't do any searches or filters. I talk to people, people comment on my blog, I email with blog readers and friends. Among all that I hear a lot of stocks and I just try to spend my stock time looking into what I've heard about
Thank you for the reply!
DeleteIt seems that this is the first nanocap "index" to be published. According to the page, stocks are selected "based on an analysis of their financial data, ownership, insider transactions, company history, stock liquidity, and (to a lesser degree) technical analysis". I guess this could be an interesting basis for finding stocks.
ReplyDeletesee here: https://www.nanocap100.com/
hello
ReplyDeleteI have tried to buy IVRO shares in Interactive Broker but it is not possible. Do you know what it is due to?
You should ask them. Trading should be allowed because they are fully reporting and in the Pink Current Information tier at otcmarkets. https://www.otcmarkets.com/stock/IVRO/overview
DeleteSomeone else told me in Sept 2022 Interactive Brokers wouldn't allow them to buy IVRO due to an "upcoming or recent corporate action". Something is messed up with that
Following up on this pullback - how does this look now, Dan, anyone, buehler?
ReplyDeleteI hold all my shares. Situation is the same as it has been to me. Lot of volume the other day which finally got rid of the large block someone had been trying to sell for some time.
DeleteMind expanding on melting ice cubes and what would make you define it as such?
ReplyDeleteWhen I say that, I just mean a business on the decline. It's not growing anymore but rather is shrinking and looks like that will continue.
Deletewould you think about selling any around 10c?
ReplyDeleteno way
ReplyDeleteI think you've convinced me Dan, I really like some of your other picks as well, I was skeptical on this one but took the time to cross-reference their financials from five different sources and then actually learn about their flagship product Corrositex and think it's brilliant. In addition, to see you're still confident they are undervalued as of 4 days ago is encouraging.
ReplyDeleteI don't know if undervalued is how I would describe IVRO. I mean at a share price of $0.09 it's trading for like 3x revenue, 16x EPS, 1.3x book value.
DeleteThe way I think about IVRO, and a lot of these tiny sleepy stocks, is potential. Market cap is only $1.8m which is super tiny. The balance sheet and share structure are tight. They have a very interesting business I can imagine taking off. Stock is low on the chart and has been building a base since forever. It shot up to $0.40-0.50 a couple years ago. I just feel like this is the sort of thing that could move up 10x, 20x, etc and so I am not selling for pennies. I am looking for a sale in the dollars.
of course it may never get that high and I could continue holding forever in the pennies. we'll see
DeleteDo you still own and has your thesis changed?
Deleteyes I still own all my shares. nothing has changed
DeleteI think there is legislation that at some point could mandate non-animal testing for cosmetics etc. That would be a home run for them. Obviously, this is not a priority for Congress at this point, but a newspaper article or TV expose about the cruelty inflicted on animals by the current testing methods could instantly propel this stock.
ReplyDeleteThank you Dan for your work and analysis and conviction. Are there any competitor to them locally or globally you are aware of and whats the size of this market if you are aware of?
ReplyDeletecompetition and market size I don't know
Deletelot of volume recently pushing this one up. Hopefully some good news coming
ReplyDeleteI would say it's due to most recent PR but that was over a month ago. https://www.otcmarkets.com/stock/IVRO/news/Game-Changing-Signals-from-Global-Regulatory-Agencies-Accompany-IVRO-Sales-and-Profit-Gains?id=503208